
Precision Microbiology for Regulated Industries
BioTrace operates as a specialized microbiological testing laboratory delivering accurate and defensible data for food, nutraceutical, herbal, cosmetic, and agricultural products. Our programs help manufacturers verify hygiene, detect contamination risks, and meet regulatory expectations with confidence.
- Validated methods with full traceability
- Accepted during audits & regulatory inspections
- Fast turnaround with structured COA reporting
Comprehensive Microbiological Testing Services
From routine enumeration to pathogen detection, our programs cover every critical microbial risk across your entire supply chain.

Total Plate Count (TPC) Analysis
Routine EnumerationTPC estimates the total viable aerobic bacteria in a product — the gold-standard indicator of manufacturing cleanliness across food, dairy, beverages, pharmaceuticals, and cosmetics. Routine TPC evaluation helps identify process deviations before they become costly batch failures.
Reveals microbial exposure from unsafe water, low-grade inputs, or inadequate sanitation practices.
Elevated counts signal increased spoilage probability and potential safety risks.
Controlled loads strengthen confidence in product stability and expected shelf life.
Trending results across batches helps optimize processing and identify hygiene gaps early.

Yeast & Mold Detection
Fungal ContaminationIdentifies fungal organisms responsible for spoilage in fruits, vegetables, spices, and plant-derived materials. Critical for product stability, preservation strategies, and long-term storage reliability while detecting contaminated inputs before they compromise entire batches.
Prevents mycotoxin risks and protects consumers from long-term health implications.
Visible discoloration, dark spots, or unusual odors suggest active fungal growth.
Validates product acceptability and understanding of humidity and packaging risk factors.
Identifies contamination root causes and supports batch-wise shelf life validation.
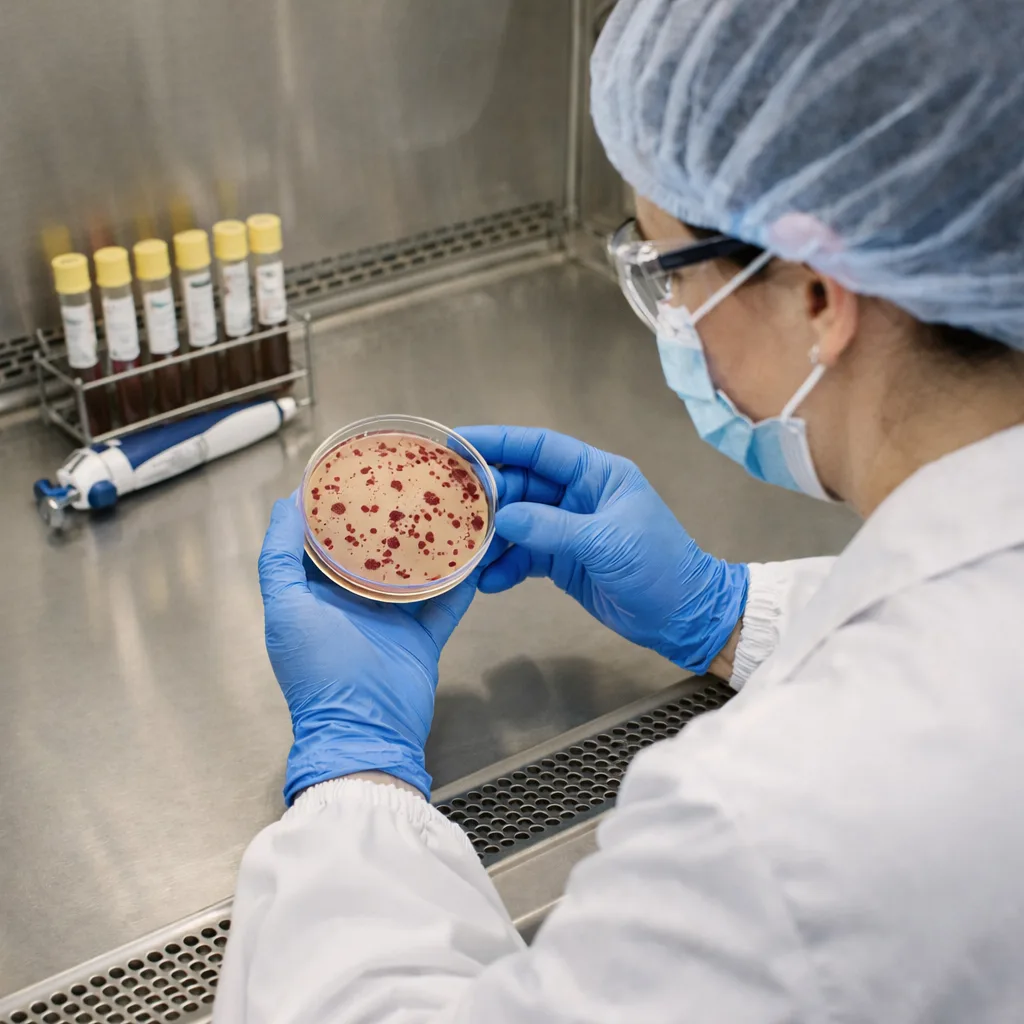
Pathogen Screening
— E. coli & Salmonella
Zero Tolerance Panel
Detects harmful organisms that can lead to serious foodborne illness and regulatory action. Fundamental for preventive control programs, export readiness, and brand protection. Our testing panel covers all critical food safety pathogens routinely required for regulatory compliance and export eligibility.
Early detection reduces recall exposure and safeguards public health.
Surface slime, abnormal moisture, or package integrity issues may signal microbial activity.
Validated pathogen testing ensures products meet safety expectations across all buyer categories.
Reports enable manufacturers to strengthen preventive systems and implement corrective measures.

Coliform Analysis
Hygiene IndicatorA key hygiene indicator reflecting contamination from water sources, equipment, or handling environments. Especially critical for fresh produce, minimally processed foods, and ready-to-use materials where sanitation directly determines product safety and overall shelf stability.
Determines sanitation effectiveness and enables early intervention before quality deteriorates.
Sticky textures, slime formation, or improper temperature control indicate elevated contamination risk.
Confirms whether hygiene controls are functioning as intended within the production environment.
Supports safer water usage, improved cleaning protocols, and stronger contamination control strategies.

Regulatory-Compliant Microbiological Testing
FDA & FSSAI AlignedBioTrace conducts testing in accordance with recognized FDA and FSSAI frameworks relevant to manufacturing, retail, and export supply chains. Strict microbial limits, standardized methodologies, and structured documentation help organizations demonstrate full due diligence and product integrity.
Regulatory compliance is essential for market access, certification, and brand protection.
Structured COAs integrate smoothly into audit and inspection environments worldwide.
Documented compliance improves buyer trust and significantly reduces approval delays.
Frequently Asked Questions
Find answers to common questions about our microbiological testing services
Protect Your Brand
from Contamination Risks
Partner with BioTrace for reliable microbiological testing and regulatory-ready documentation that gives buyers and regulators full confidence.
How can I help you?
